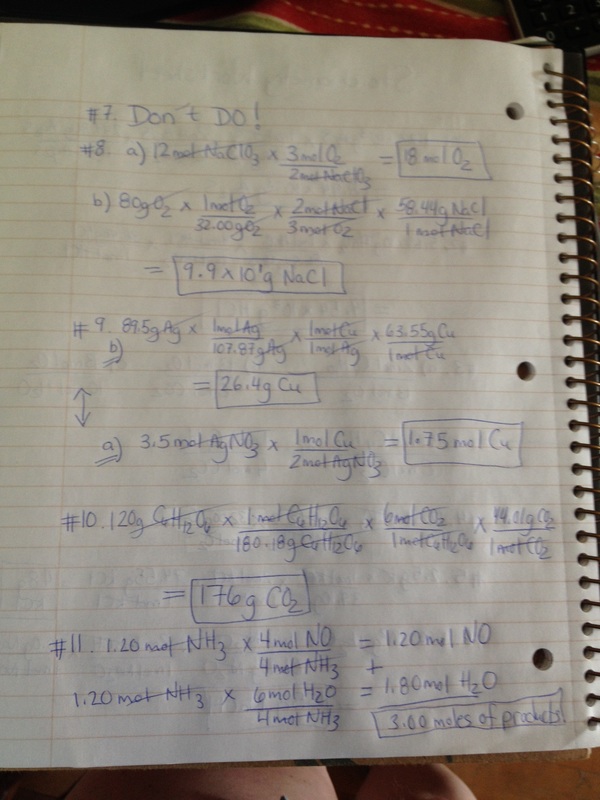September
Significant Figure Homework Sheet
| significant_figures.pdf | |
| File Size: | 377 kb |
| File Type: | |
Answers to Significant Figures Worksheet and Measurement Question 1. a) 5 b) 4 c) 1 d) 6 e) 3 f) 6 g) 4 h) 5 i) 5 j) 3 k) 1 l) 6 2. a) 169.0 b) 582.8 c) 2026.8 3. a) 9.0 x 10^1 b) 11 c) 0.81 d) 417 e) 2.80 x 10^3 f) 4.0 g) 7.58 h) 4252 i) 201 j) 1.1 x 10^3 k) 159 4. Syringe A - 15 ml + 5 ml Syringe B - 15.0 ml + 0.5 ml
Mole Worksheet/Answers
| moles_molecules_grams_worksheet_ans.doc | |
| File Size: | 25 kb |
| File Type: | doc |
Stoich Answers
Quantum
Chemistry textbook:
pg 51 # 4 (refer to pg. 372 for hazard symbols), #5, #15, #18, #20
pg 62 # 10, # 11
pg 68 # 3, # 5, # 7, # 8, # 9
Answers to textbook:
pg
51 #4 a) The product must be kept away from heat and flames, and the container
must be perforated.
b) Contact with skin and eyes must be avoided. Safety glasses, gloves and a
lab coat or apron must be worn.
c) The product must be kept away from heat, flames and sparks.
#5 a) 3 b) 4 c) 2 d) 1
#15 At high altitudes in the atmosphere, the temperature is very low and water
vapour released by jet engines condenses into ice crystals.
#18 Carbon dioxide, a greenhouse gas is taken from the atmosphere for the
photosynthesis reaction.
#20 Hydrogen is a flammable gas that caused the loss of the zeppelin when it caught
fire in an accident. Helium is an inert gas and would not have burned in
that way if it had been used to inflate the balloon.
pg 62 #10 b) Gas particles are not attracted to each other, because there is no
force of attraction or repulsion between them.
#11 a) The mean distance between the particles of a gas is significantly
greater than the size of the particles. It is therefore possible to
compress gases and bring their particles closer together. The smaller
space between the particles of liquids makes it impossible to compress
them.
b) The particles of a gas are extremely far apart. Thus the number of
particles per unit volume is smaller; that is, the density is lower.
c) Since the particles of a gas are completely independent of each other and move
continuously and in a straight line in all directions, the volume of a gas
varies according to the space available.
d) The particles of a liquid pile up on each other, because they are drawn to each
other by a force of attraction, while particles of a gas are very far apart
from each other. Thus the liquid always occupies the same amount of space,
while gas particles fill all of the space of the container they are
in.
e) At a given temperature, the mean kinetic energy of gas particles is the same for
all gases. SInce the kinetic energy of a gas particle in motion is found
by using the formula Ek=1/2 mv2 , at a given temperature, the
velocity of gases with high mass is less than those with low mass. The
particles of nitrogen are heavier than those of hydrogen (molar mass
nitrogen = 28.02 g/mol and hydrogen 2.02 g/mol). Therefore the
particles of nitrogen move more slowly.
pg 68 #3 The compressibility of gases allows the hatchbacks to absorb the
shock of striking the body of the car.
#5 The particles move continuously in a straight line in all directions. Since
gases have no defined shape or volume, they dilate indefinetely by filling any
available space. When the balloon rises, atmospheric pressure
decreases and the helium in the balloon expands. The expansion of the
helium stretches the surface of the balloon, and the helium occupies a greater
volume. That is why the size of the balloon increases.
#7 NH3, HF, CO, NO2, HI
#8 The molar mass of the unknown gas is 24.4 g/mol.
#9 The molar mass of the unknown gas is 146 g/mol.
Challenging Problems for diffusion and KMT with answers see attached worksheets
below
below
| kmt_and_grahams_law.doc | |
| File Size: | 27 kb |
| File Type: | doc |
| kmt_and_grahams_law_answers.doc | |
| File Size: | 30 kb |
| File Type: | doc |
Pressure (p.74 #1-2)
1. When particles collide with the side of the container they exert a force.
Pressure is measured as the force per unit area.
2. 688 mmHg
a) 0.905 atm
b) 91.7 kPa
1. When particles collide with the side of the container they exert a force.
Pressure is measured as the force per unit area.
2. 688 mmHg
a) 0.905 atm
b) 91.7 kPa